
The efficiency of uranium recovery operations largely depends on one critical decision: selecting the right ion exchange resin. In modern uranium mining—whether in conventional acid leaching circuits or in-situ leaching (ISL) operations—the difference between strong base anion (SBA) and weak base anion (WBA) resins can significantly impact recovery rates, resin life cycle, regeneration cost, and overall operational profitability.
This guide provides a detailed comparison of SBA and WBA resins for uranium extraction and explains how to determine which resin type best fits your mining conditions.
Understanding Uranium Chemistry in Leach Solutions
Before selecting a resin, it is essential to understand the chemical form of uranium in the leach liquor.
In sulfuric acid leaching systems—commonly used in uranium mining—uranium exists primarily as negatively charged sulfate complexes such as:
UO₂(SO₄)₃⁴⁻
UO₂(SO₄)₂²⁻
Because these complexes are anionic, anion exchange resins are required for effective recovery.
However, leach solutions rarely contain uranium alone. They also include competing ions such as:
Ferric iron (Fe³⁺)
Chloride (Cl⁻)
Silica
Organic contaminants
These components influence resin selection, fouling tendency, and regeneration efficiency. Therefore, understanding solution chemistry is the foundation of selecting the correct uranium extraction resin.
Strong Base Anion (SBA) Resins in Uranium Recovery
What Is a Strong Base Anion Resin?
Strong base anion resins contain quaternary ammonium functional groups. These groups remain fully ionized across the entire pH range (0–14), allowing the resin to operate in both strongly acidic and alkaline conditions.
In uranium extraction systems—especially sulfuric acid leach circuits—SBA resins demonstrate strong electrostatic attraction toward uranium sulfate complexes.
Performance Advantages of SBA Resins
High Uranium Selectivity
SBA resins offer excellent selectivity for uranium sulfate complexes, even in high sulfate environments.
Wide Operating pH Range
Because they remain active across all pH levels, they provide operational flexibility.
Stability in Strong Acid Systems
In highly acidic leach liquors, SBA resins maintain structural and chemical integrity.
Proven Performance in ISL Operations
Many in-situ leaching uranium projects rely on SBA resins due to their predictable and stable performance.
Limitations of SBA Resins
Despite their strong performance, SBA resins have certain drawbacks:
Higher regeneration chemical consumption
Greater susceptibility to organic fouling
Higher initial cost compared to WBA resins
In operations with heavy organic contamination or lower acid strength, SBA may not always be the most cost-effective choice.
Weak Base Anion (WBA) Resins in Uranium Extraction
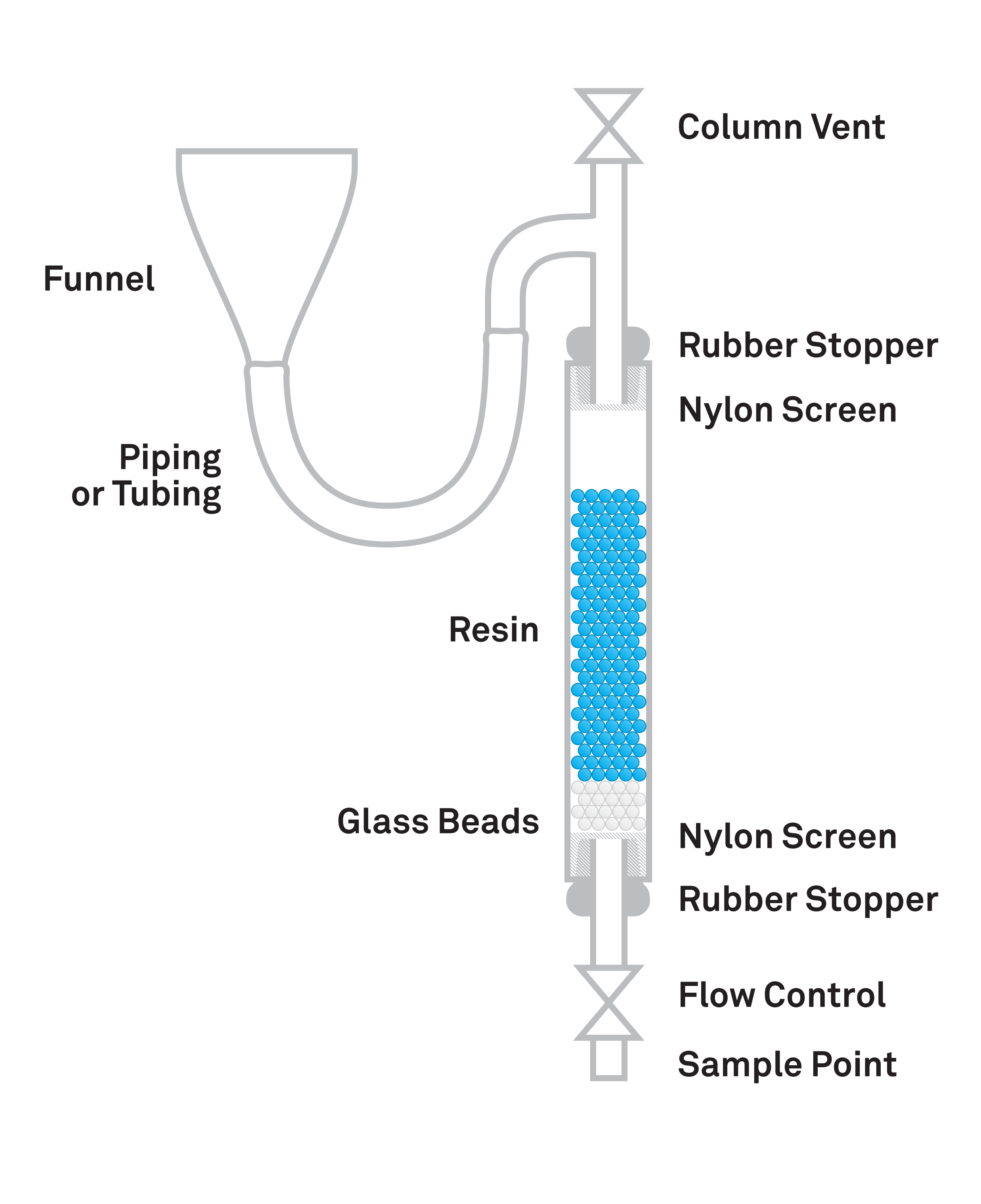
What Is a Weak Base Anion Resin?
Weak base anion resins contain tertiary amine functional groups. Unlike SBA resins, WBA resins are only ionized and active under acidic conditions.
They do not function effectively in neutral or alkaline environments, but under properly controlled acid systems, they can offer significant economic advantages.
Performance Advantages of WBA Resins
Lower Regeneration Chemical Demand
WBA resins generally require less caustic soda or regenerant chemicals, reducing operational cost.
Better Organic Fouling Resistance
They tend to exhibit stronger resistance to organic contamination, extending resin life in challenging environments.
Lower Operating Cost
In moderate acid systems, WBA resins can reduce total operating expenses.
Simplified Regeneration Cycles
Because of their functional group chemistry, regeneration is often easier and more economical.
Limitations of WBA Resins
However, WBA resins are not suitable for every uranium recovery system:
Limited pH operating range
Reduced performance in very high sulfate concentration
Not ideal for strongly acidic leach circuits
When sulfate levels are extremely high, SBA resins typically outperform WBA alternatives.
Direct Comparison: SBA vs. WBA for Uranium Mining
| Parameter | SBA Resin | WBA Resin |
|---|---|---|
| Functional Group | Quaternary Ammonium | Tertiary Amine |
| Operating pH | 0–14 | Acidic Only |
| Uranium Selectivity | High | Moderate–High |
| Sulfate Tolerance | Excellent | Moderate |
| Organic Resistance | Moderate | High |
| Regeneration Cost | Medium–High | Low |
| Typical Use | Strong acid systems | Moderate acid systems |
There is no universal “best” uranium extraction resin. The optimal choice depends entirely on your process chemistry and economic model.
How to Select the Right Uranium Extraction Resin
Selecting the correct resin requires systematic evaluation rather than simple price comparison.
Analyze Leach Liquor Chemistry
Determine:
Sulfate concentration
Ferric iron loading
Organic contamination
Total dissolved solids
Without accurate chemical analysis, resin selection becomes guesswork.
Evaluate Operating Conditions
Consider:
pH range
Flow rate
Temperature
Bed depth and column design
Dynamic operating conditions significantly influence breakthrough performance.
Compare Life-Cycle Cost (Not Just Resin Price)
True cost includes:
Resin lifespan
Regeneration chemicals
Downtime risk
Replacement frequency
Often, a higher-priced resin with longer service life results in lower total cost per pound of recovered uranium.
Conduct Pilot Column Testing
Laboratory batch tests are not sufficient. Column testing should evaluate:
Dynamic exchange capacity
Breakthrough curves
Pressure drop
Mechanical stability
Pilot testing provides realistic performance data under actual flow conditions.
Common Mistakes in Uranium Resin Selection
Many uranium recovery projects face performance issues due to avoidable mistakes:
Selecting resin solely based on price
Ignoring iron fouling potential
Underestimating organic contamination
Failing to evaluate regeneration efficiency
Not performing pilot column validation
A data-driven selection process prevents long-term operational inefficiencies.
Conclusion
In uranium extraction, resin selection is not a minor procurement decision—it is a strategic operational factor.
Strong base anion resins offer superior performance in high acid and high sulfate systems. Weak base anion resins provide cost advantages and better organic resistance in moderate acid conditions.
The correct choice depends on:
Leach solution chemistry
Process conditions
Regeneration strategy
Long-term economic evaluation
Mining operations that adopt a performance-first, data-driven resin selection approach consistently achieve:
Higher uranium recovery rates
Longer resin service life
Lower chemical consumption
Reduced operational cost
If you are evaluating ion exchange resins for uranium recovery, technical consultation and pilot testing are essential steps to ensure optimal performance.




